Solving Non-Invasive Glucose Monitoring
We’re excited to announce a major breakthrough toward non-invasive glucose monitoring, the Holy Grail of self-measurement.
Despite billions of dollars and decades of research, non-invasive glucose monitoring remains an unsolved problem. Nearly 10% of the world population lives with diabetes and must constantly measure their blood glucose levels to manage their condition. Today, every measure of glucose is invasive, with the vast majority of people relying on finger-pricks. This is a massive health crisis responsible for the daily suffering of hundreds of millions of people.
Work toward non-invasive glucose monitoring began in the early 1970s, and since then numerous techniques have been explored, including infrared absorption spectroscopy, Raman spectroscopy, photoacoustic spectroscopy, transdermal techniques like reverse iontophoresis, and correlation with other biofluids such as sweat or saliva, among others. But after decades of research, there has yet to be an accurate, successful technology.
This body of failures is represented in one sentence: no portable technique has ever reliably been shown to non-invasively and directly measure a unique signal specific to the glucose molecule in vivo at relevant physiological concentrations.
Today, we are excited to announce a major breakthrough that establishes a path toward the first accurate non-invasive glucose monitor capable of finally solving this decades-long challenge.
For the first time, we have demonstrated that we can directly measure glucose non-invasively using magnetic resonance spectroscopy.
Magnetic resonance spectroscopy (MRS) is a well established technique dating back to the 1950s. The underlying principle is similar to MRI, but rather than producing images, MRS produces spectra that allows us to observe and quantify different molecules non-invasively. In the human body, MRS allows for the direct measurement of metabolism in different organs and tissues without worrying about penetration depth challenges that other spectroscopies have struggled to overcome.
How it works
Every hydrogen atom has one proton in its nucleus. This proton produces a tiny magnetic field measured as a magnetic moment. When placed in an external magnetic field and a radiofrequency pulse is applied, these protons will resonate at a very specific frequency called the Larmor frequency. In a given molecule, protons, influenced by local electrons, will resonate at slightly different frequencies, allowing us to differentiate molecules by measuring the frequencies of these hydrogen signals (in units of parts-per-million or ppm).
As an example, the proton magnetic resonance spectrum of a mixture of two molecules, glucose and lactate, is shown below:
Two main groups of signals are observed that correspond to the two different molecules, with the majority of glucose on the left resonating between 3 and 4ppm (blue), and lactate resonating at about 1.3ppm (orange). The corresponding hydrogen atoms that produce these signals are highlighted in blue and orange on the molecular diagrams.
The important point to emphasize here is that the area under these signals is directly proportional to the amount of the molecule that is being measured. This is how we can quantify the concentration of these molecules non-invasively.
Similar to MRI scanners, magnetic resonance spectrometers generally require very powerful magnetic fields and room-sized equipment. A typical spectrometer is shown below:
Though the science is powerful, making this technology practical as a low-cost, small-scale device is very challenging.
Our key hypothesis has been to make MRS measurements using weak magnetic fields instead of the very strong, commonly used field strengths. This allows us to highly miniaturize the entire system by many orders of magnitude.
Our breakthrough
Using a low-field magnet, we miniaturized this technology into a small and portable tabletop form-factor that can sit on someone’s desk. A user inserts their finger into the device and we make measurements at their fingertip.
Only two things are used to make a measurement: a magnetic field and low-frequency radio waves. So the overall power deposited is significantly less than a typical smartphone.
This miniaturization of a magnetic resonance system is unprecedented.
For the first time, we’ve shown that we can directly measure blood-glucose non-invasively using this approach. This is not a correlation with glucose – this is a direct measurement of the glucose molecule itself.
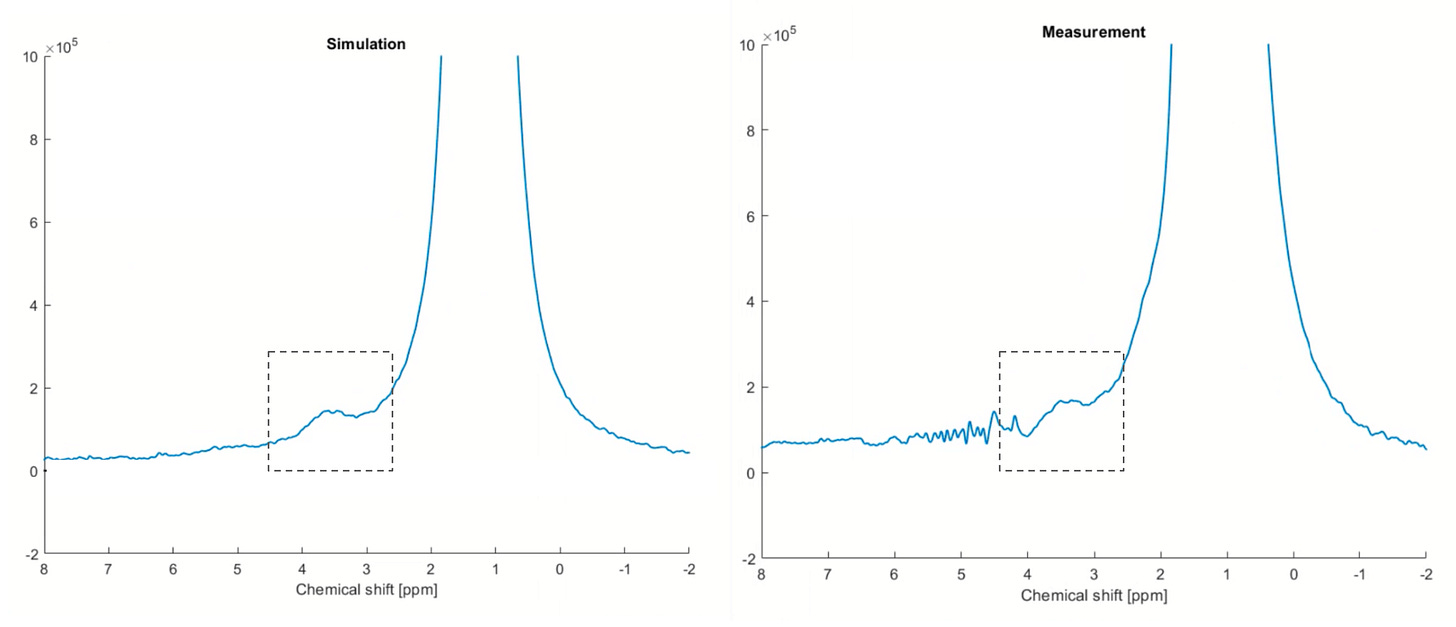
The result of an example finger measurement is shown below (right) compared against the prediction from simulation (left). The glucose signal is highlighted in the dashed box between 3 and 4ppm. The measurement matches the prediction extremely well. Note the much larger signal on the right of glucose coming from the lipids in the finger. The dominant water signal at 4.7ppm, which is present almost everywhere in the human body, was digitally removed to better illustrate the glucose detection.
We verified that this signal was reproducible in different people and results were not impacted by the limitations observed in other spectroscopies, including skin color. Importantly, we’ve shown that the area under this signal correlates strongly with blood-glucose concentration. In a preliminary regression analysis, we were able to achieve an R-squared of 0.95 compared to a reference finger-prick device:
No machine learning was used in this data analysis, so there is no training dataset or patient calibration, unlike almost every other approach that has been attempted. Because we can directly measure glucose itself, we can achieve unprecedented accuracy without the need for deeply complex quantification algorithms.
This result is a massive breakthrough and highlights the path toward the first accurate, non-invasive glucose monitor.
We are incredibly excited to be sharing this result with the world. This breakthrough is the culmination of years of work across many disciplines that includes materials science, electromagnetics design, digital electronics, magnetic resonance physics, digital signal processing, and more.
Our results have been peer-reviewed by three leading independent experts in magnetic resonance. Two quotes from our reviewers are included below:
“What Synex has done is the biggest breakthrough in non-invasive health monitoring that I’ve seen in recent years. Showing that a portable magnetic resonance system can directly measure glucose is groundbreaking, and what many thought to be impossible. Their data is compelling, and highlights a path to a world-changing product with the potential to measure important metabolites in vivo.”
Dr. Matt Rosen, Director of Low-Field MRI at Harvard and Mass General Hospital
“Magnetic resonance is the most versatile spectroscopic technique and allows for the direct measurement of dozens of different metabolites in vivo. By miniaturizing to such a small scale, Synex has shown that it's possible to directly observe metabolism in a portable form factor, paving the way to a new generation of health monitors.”
Dr. Andre Simpson, Director of Environmental NMR Spectroscopy at University of Toronto
A vision of the future
We are hard at work turning this breakthrough into a world-changing product. Across the world, people with diabetes have to bear the pain of invasive measurements every day. We hope that this technology can finally put an end to this.
Our vision of the future goes beyond glucose. As a versatile technique, magnetic resonance spectroscopy has the capability to measure many more metabolites than glucose, including lactate, ketones and amino acids, all with the same hardware.
We strongly believe that the future of healthcare must be proactive. Today, wearables and consumer health sensors are restricted to measuring basic vital signs like heart rate and blood oxygen. Our technology has the capability to take this to the next level by giving people access to their metabolism to help manage dozens of conditions and finally deliver a future of predictive health.
The future of health monitoring is coming.






Please send wire details
Dear colleagues. Our research group is very close to decide of the non-invasive sugar measuring problem. We can send a high level description of our project because very interest to cooperation with Toronto scientific research's. Will be glad to our furthermore contacts.
With best wishes Dr. Victor Kardashov, PhD, main inventor of the computer assisted blood sugar level p[program.